Many people have difficulty when switching to a waterborne finishing system. I think this is because they approach it like they would a solvent-based system, which will give mixed results at best. The first rule to remember when making the switch is that you are dealing with a water emulsion rather than a solvent solution so the entire chemistry dictates a different set of rules that you have to follow. With a better understanding of the fundamentals and how they relate to application techniques, it will make the transition easier and less frustrating.
Waterborne Finishes
Our discussion will center on waterborne finishes, and not water-based finishes. Water-based finishes are essentially latex paint without the pigment. While there are certain applications for water-based finishes, most furniture and cabinetry products are better suited for waterborne finishes.
To understand waterborne finishes a bit better, let’s look at solvent-based finishes first.
- Resins
- True or Active Solvents
- Diluent Solvents
- Latent Solvents
Basic Solvent based Finish Components:

Various resins, both man-made and natural, are selected for their particular performance characteristics: hardness, flexibility, scratch resistance, water resistance, and cost. Mixed in the proper ratio, they will give the desired properties for the finish.
There are three classes of solvents in a finish, and each class contains multiple solvents.
True solvents are solvents that will melt the resins into a liquid at room temperature. When added to the resins, they melt into a thick syrupy liquid that is too thick for spray application. Diluent solvents do not melt resins at room temperature. However, they do mix with the True solvents, reducing the viscosity of the resin-solvent mixture to a level that gives it the ability to be sprayed.
Latent solvents are solvents that, on their own, will not melt resins at room temperature. Instead, they are “activated” by the True solvents, increasing their potency. They are also less expensive than True solvents, thus reducing the overall cost of the finish.
Once mixed, the resins and solvents form a solution. This means that the resin is completely dissolved in the solvent, just like salt dissolves in water. When the finish is sprayed onto the wood, the solvents begin to evaporate. Each of the solvents evaporate at different rates. The solvents that evaporate faster, generally the diluent and some latent solvents, are the ones that “thin out” the finish so it can flow onto the wood. As these evaporate, the slower solvents (usually the True solvents) are left to melt the resins into a smooth thin film. Although temperature and humidity affect how fast this all happens, the whole process takes place in a relatively short period of time.
- Polymer Emulsion – Acrylic or Polyurethane
- Coalescent Solvent – usually a Glycol Ether
- Water
Basic Waterborne Components:

There are several polymer formulations, including acrylic, acrylic-polyurethane, polyurethane and acrylic, and finally straight polyurethane. The polymers are combined to take advantage of the non-yellowing characteristics of the acrylic and the increased wear characteristics of the poly. Obviously, the greater the amounts of polyurethane present, the harder the finish, but also the chance of yellowing increases.
 This sounds a little odd, but water-based finishes are actually formulated on a solvent-based platform. Glycol ether solvent is used because it is compatible with water, and will melt most acrylic and polyurethane resins. Another factor is that it evaporates more slowly than water. It importance of this will be shown later.
This sounds a little odd, but water-based finishes are actually formulated on a solvent-based platform. Glycol ether solvent is used because it is compatible with water, and will melt most acrylic and polyurethane resins. Another factor is that it evaporates more slowly than water. It importance of this will be shown later.
The basic waterborne finish can be defined fairly simply; Polymer resins are manipulated to form tiny droplets called latexes. They are dispersed in the water to create an emulsion. An emulsion is a liquid made up of unblendable substances. A good example of an emulsion is oil and vinegar. Then the coalescent solvent is added. Even though the process is relatively simple to explain, the chemistry involved is another matter. The polymers and water are not chemically compatible, so getting them to work together is quite complicated. Overcoming the issues of surface tension, ph, grain raise, and foaming are much more difficult than in their solvent-based cousins.
A simple explanation for the drying process is that the water evaporates and the polymers form a layer on the surface of the wood. At this point, the coalescent solvent is at a high enough concentration to dissolve or soften the polymers so that they melt together forming a thin smooth film on the wood.
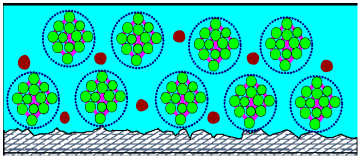
Polymers and coalescing solvent in water emulsion

As water evaporates the concentration of coalescing solvent increases
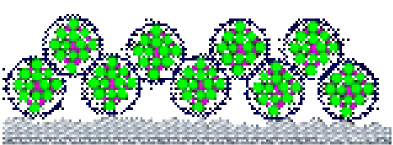
As the coalescing solvent evaporates, it melts the polymer to form a flat film
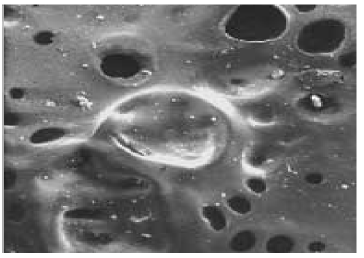
Finish right after spraying. Holes are atomizing air and solvents are evaporating
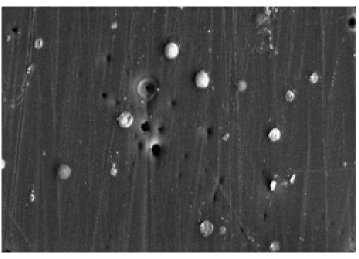
Dried finish. Notice pin holes and sanding scratches. White dots are trapped air bubbles

Cross section of dry finish. The blue arrow shows the sealer coat while the red arrow shows the top coat.
Waterborne finishes produce a coating that has excellent water and abrasion resistance. However, their Achilles Heel is solvent resistance. Remember that a coalescing solvent is used to melt the resins together when they dry. Although they are not yet up to the caliber of the best-catalyzed solvent-based finishes, the difference is becoming smaller with the introduction of pre and post-catalyzed waterborne products.
Manufacturers are currently working on a couple of issues that are keeping waterborne from becoming the standard in the industry. Clear waterborne finishes are almost perfectly clear. Also, they lay on the surface a little more than solvent-based finishes, so they tend to brighten the natural variations in the wood. This can result in a slightly more harsh or plastic look, although they are getting much better. Solvent-based finishes tend to soften these variations, resulting in a warmer look. Waterborne finishes are also are more weather-sensitive, but this can be overcome with application techniques. The sophisticated chemistry can add to the cost of waterborne finishes, but never use the dollar-per-gallon cost as your main criteria for the acceptability of a finish. Tighter health, environmental, and safety regulations are fostering an increase in waterborne research and use.
